- a packaging signal (P) needed for the assembly of fresh virus particles
- inverted repeats ("R") at each end; to aid insertion of the DNA copies into the DNA of the target cell.
| Index to this page |
Many human diseases are caused by defective genes.
A few common examples:| Disease | Genetic defect |
|---|---|
| hemophilia A | absence of clotting factor VIII |
| cystic fibrosis | defective chloride channel protein |
| muscular dystrophy | defective muscle protein (dystrophin) |
| sickle-cell disease | defective beta globin |
| hemophilia B | absence of clotting factor IX |
| severe combined immunodeficiency (SCID) | any one of several genes fail to make a protein essential for T and B cell function |
All of these diseases are caused by a defect at a single gene locus. (The inheritance is recessive so both the maternal and paternal copies of the gene must be defective.) Is there any hope of introducing functioning genes into these patients to correct their disorder? Probably.
Other diseases, also have a genetic basis, but it appears that several genes must act in concert to produce the disease phenotype. The prospects of gene therapy in these cases seems far more remote.
It is a disease of young children because, until recently, the absence of an immune system left them prey to infections that ultimately killed them.
About 25% of the cases of SCID are the result of the child being homozygous for defective genes encoding the enzyme adenosine deaminase (ADA). The normal catabolism of purines is deficient, and this is particularly toxic for T cells and B cells.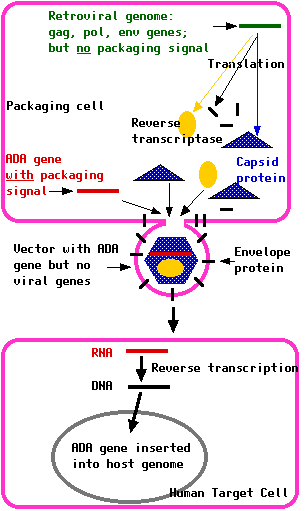 Treated with these two genomes, the packaging cell produces a crop of retroviruses with:
Treated with these two genomes, the packaging cell produces a crop of retroviruses with:Once the virus has infected the target cells, this RNA is reverse transcribed into DNA and inserted into the chromosomal DNA of the host.
| External Link |
| View an animation showing the steps used in retroviral gene therapy at this Molecular Medicine in Action site. Navigate to Other → Viruses as Vectors on the main menu. (You need Flash 6.) |
| Please let me know by e-mail if you find a broken link in my pages.) |
The first attempts at gene therapy for SCID children (in 1990), used their own T cells (produced following ADA-PEG therapy) as the target cells.
The T cells were:| But someday there may be a way to make genetically-engineered T cells live indefinitely by getting them to express telomerase. See the discussion. |
In 1993, ADA gene therapy was attempted with blood ("hematopoietic") stem cells drained from the umbilical cords of three newborn babies who were known to be homozygous for ADA deficiency.
Blood stem cells:| Discussion of blood stem cells |
At last report, some of the circulating T cells in these children carried the ADA gene. But because the children continue to receive ADA-PEG, it is unclear how much gene therapy has contributed to their good health.
However, in June of 2002, a team of Italian and Israeli doctors reported on two young SCID patients that were treated with their own blood stem cells that had been transformed in vitro with the ADA gene. After a year, both children had fully-functioning immune systems (T, B, and NK cells) and were able to live normal lives without any need for treatment with ADA-PEG or immune globulin (IG). The doctors attribute their success to first destroying some of the bone marrow cells of their patients to "make room" for the transformed cells.Gene therapy has also succeeded for 15 French baby boys who suffered from another form of severe combined immunodeficiency, called X-linked SCID, because it is caused by a mutated X-linked gene encoding a subunit — called γc (gamma-c) — of the receptor for several interleukins, including interleukin-7 (IL-7).
IL-7 is essential for converting blood stem cells into the progenitors of T cells. [View]. Boys with X-linked SCID can make normal B cells, but because B cells need T-helper cells to function, these boys could make neither cell-mediated nor antibody-mediated immune responses and had to live in a sterile bubble before their treatment.
The French doctors*
Three of the little boys have developed cancer (one has died):
|
Link to discussions of other approaches to gene therapy that are being tried.
|
| Welcome&Next Search |